Precision-Guided Dosing

![[Page Header] IFXm 20230705 [Page Header] IFXm 20230705](https://prometheuslabs.com/wp-content/uploads/2023/07/Page-Header-IFXm_20230705.png)
Conventional approaches to drug monitoring can lead to unoptimized therapy
resulting in inefficient care, disease recurrence and potential treatment failure
Anti-TNFs have revolutionized the treatment and management of inflammatory bowel diseases (IBD) but still present challenges. Roughly 50% of initial responders will experience secondary loss of response within the first year of treatment, often attributed to subtherapeutic biologic concentrations.1
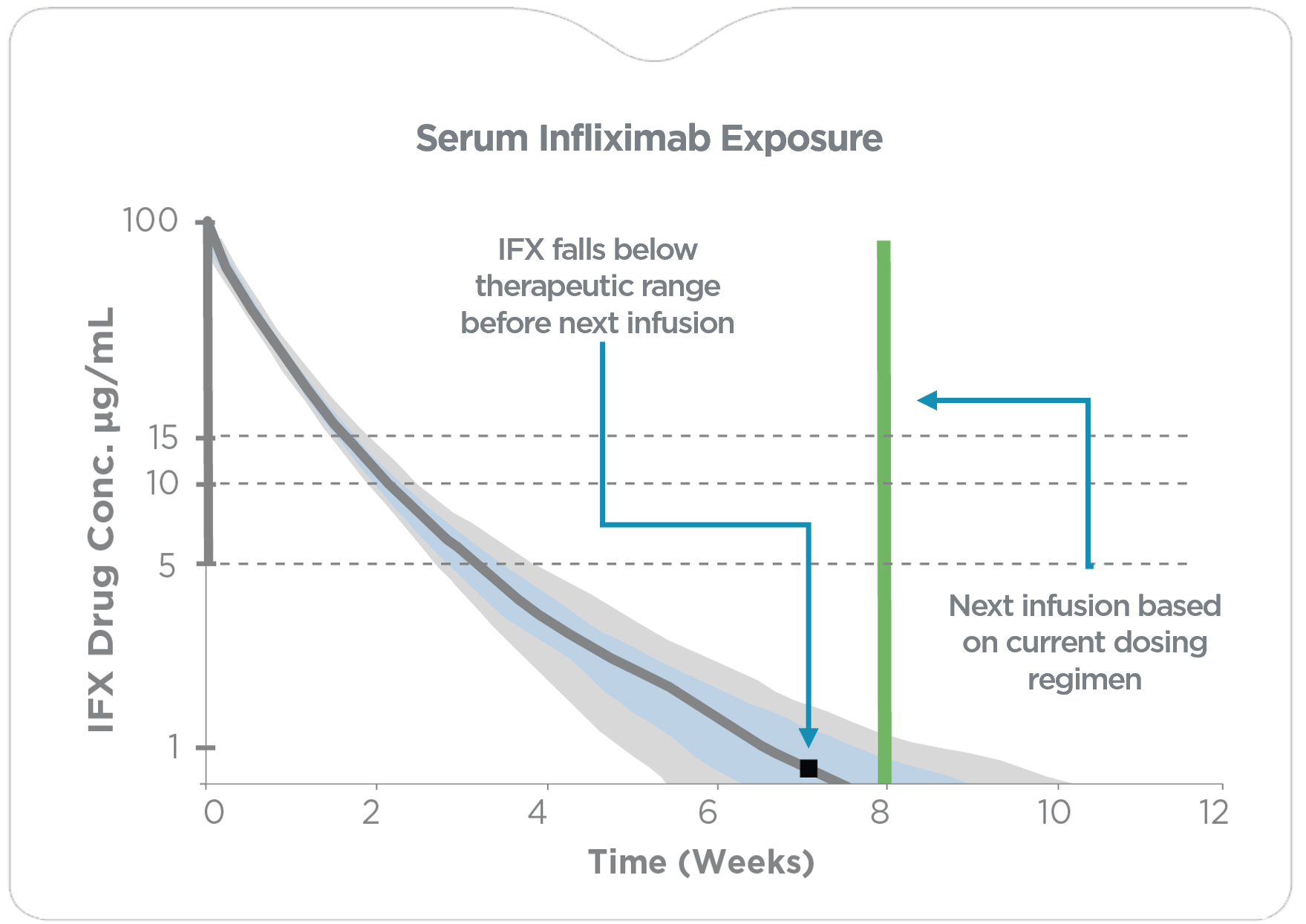
Current standard of care involves a cycle of treatment, reactive therapeutic drug monitoring (TDM) and empiric dose adjustments. No actionable guidance on how to expediently escalate or de-escalate dosing to achieve target concentrations is available.
What if your patient’s unique pharmacokinetic profile could guide their treatment, help mitigate treatment failures, increase IFX durability and improve remission rates?
The cycle of ad hoc dose adjustments puts an unnecessary strain on IBD patients
![[Chart] Section 2 – Estaimted IFX Table_20230314 [Chart] Section 2 - Estaimted IFX Table_20230314](/wp-content/uploads/2023/03/Chart-Section-2-Estaimted-IFX-Table_20230314.png)
PredictrPK IFX testing during maintenance quickly assess a patient’s pharmacokinetic (PK) profile to facilitate individualized precision-dosing of IFX. The test offers actionable insights with projected serum IFX concentrations based on the current dosing regimen as well as with alternative dosing options.
Precision-guided dosing with PredictrPK increases the likelihood of achieving therapeutic targets, recapturing clinical response and shortening the time to remission.
PredictrPK provides forward looking insights to precisely optimize IFX therapy
Precision-guided IFX dosing during maintenance supports durable drug response and optimal dosing
![[Chart] Section 3 – Likelihood of Clinical Remission by Est IFX_20230314 [Chart] Section 3 - Likelihood of Clinical Remission by Est IFX_20230314](/wp-content/uploads/2023/03/Chart-Section-3-Likelihood-of-Clinical-Remission-by-Est-IFX_20230314.png)
![[Line Graph] Section 3 – Forecasted vs Measured IFX_20230314 [Line Graph] Section 3 - Forecasted vs Measured IFX_20230314](/wp-content/uploads/2023/03/Line-Graph-Section-3-Forecasted-vs-Measured-IFX_20230314.png)
• Estimated IFX troughs of 5-10 μg/mL, and to a greater extent ≥10 μg/mL, correlated with higher likelihood of CRP-based clinical remission (Fig. 1).2
• Excellent correlation between forecasted and observed IFX trough levels (Fig. 2; p < 0.01).2,3
PredictrPK provides actionable data, before the next scheduled infusion,
to address sub-therapeutic IFX concentrations and optimize IFX therapy earlier.
CRP-based clinical remission: CD: CRP <3 mg/L, HBI <5 and CDAI <150 points, (or PCDAI <10). UC: (UC), CRP <3 mg/L, Partial Mayo <2 and PUCAI <10.
References




