Inflammatory Bowel Disease

Proven test performance, validated by the numbers1
The Monitr® Crohn’s Disease Test features a proprietary Endoscopic Healing Index (EHI) Score—ranging from 0 to 100—that was developed and validated using endoscopy-paired serum samples from 195 adult Crohn’s disease (CD) patients from a US-based tertiary care center.
Within the Monitr validation cohort patient profile:
- 77% of patients had a history of biologic medication treatment
- Median duration of CD was over 10 years
- 46% of patients had a history of inflammatory bowel disease (IBD)-related surgery
Optimal thresholds identified1
Optimal EHI thresholds were identified to maximize sensitivity and specificity for ruling in—and ruling out—endoscopically active disease, including*:
- At an EHI cutoff of 20, the sensitivity is 83% for ruling out endoscopically active disease, with a specificity of 37% predictive value for identifying patients in remission or with mild endoscopic disease†
- At an EHI cutoff of 50, the specificity is 88% for ruling in endoscopically active disease, with a sensitivity of 30%†
- As EHI cutoffs increase from 20 to 50, specificity steadily increases, indicating a higher likelihood of active disease
EHI performance vs other biomarkers1
In the validation, EHI performance was compared to clinically relevant C-reactive protein (CRP) and fecal calprotectin (FC) cutoffs in a subcohort of 81 adult CD patients.
Sensitivity
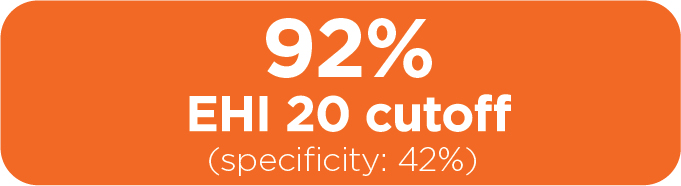
for ruling out endoscopically active disease


Specificity

for ruling in endoscopically active disease


The EHI demonstrated consistent performance across disease locations and disease behaviors.1
Additional results
In a separate and distinct cohort of 116 adult CD patients from a multisite clinical trial, EHI performance was compared to clinically relevant CRP and FC cutoffs. In this cohort patient profile:
- Patients were biologic naïve
- Median CD history was < 1 year
- 10% of patients had a history of IBD-related surgery
Sensitivity
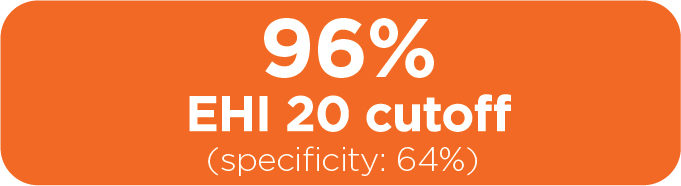
for ruling out endoscopically active disease


Specificity

for ruling in endoscopically active disease


Measure, monitor, and track endoscopic disease activity with 1 simple serum test. Get the Monitr Test Requisition Form
Need ordering assistance?
Easy to order: Monitr Test Requisition Form. For assistance ordering,
contact Client Services toll-free in the US at 888-423-5227 (Monday through Friday from 6 AM to 4:30 PM PT).
*The EHI was validated using endoscopy-paired serum samples from 195 adult CD patients from a US-based tertiary care center. Validation cohort included patients with a longer-term disease duration who may have failed more than 1 biologic or had IBD-related surgeries. The EHI Score is intended to aid in the assessment of endoscopic disease activity in adult CD patients in conjunction with clinical evaluation performed by a healthcare professional. The Monitr test is not intended to diagnose CD.
†Endoscopically active disease defined as Crohn’s Disease Endoscopic Index of Severity (CDEIS) ≥ 3, Simple Endoscopic Score for Crohn’s Disease (SES-CD) > 2, or SES-CD = 2 (if only 1 intestinal segment had a score of 2 and scores of 0 in remaining segments).
References
1. D’Haens G, Kelly O, Battat R, et al. Development and validation of a test to monitor endoscopic activity in patients with Crohn’s disease based on serum levels of proteins. Gastroenterology. 2020;158(3):515-526.e10.
